Prospective observational studies (Stewardson et al., Thyroid, 2023; Wu et al., Thyroid, 2025)
Prospective observational studies were conducted in the AHS Calgary Health Care Region, where a guidelines-based thyroid nodule pathway (PCN pathway) including thyroid nodule ultrasound malignancy risk stratification (EFN white paper) and determination of local malignancy risk for each Bethesda category (Ghaznavi et al., Acta Cytologica, 2022) has recently been implemented.
ThyroSPEC™ was prospectively validated in the AHS Calgary Health Care Region in Southern Alberta with Bethesda III (AUS/FLUS) or Bethesda IV (FN/SFN) thyroid nodules diagnosed from July 30, 2020, until July 31, 2022 (Stewardson et al., Thyroid, 2023). The integration of ThyroSPEC™ results with additional clinical variables for 1024 patients facilitated the creation of a nomogram which allows further improvement of malignancy risk prediction for patients with intermediate malignancy risk mutations and no mutation detected (Wu et al., Thyroid, 2025). The pre-test risk of malignancy was 26% in the AUS/FLUS category and 43% in the FN/SFN category prior to implementation of molecular diagnostics with respective resection rates of 21% and 56% (Ghaznavi et al., 2022).
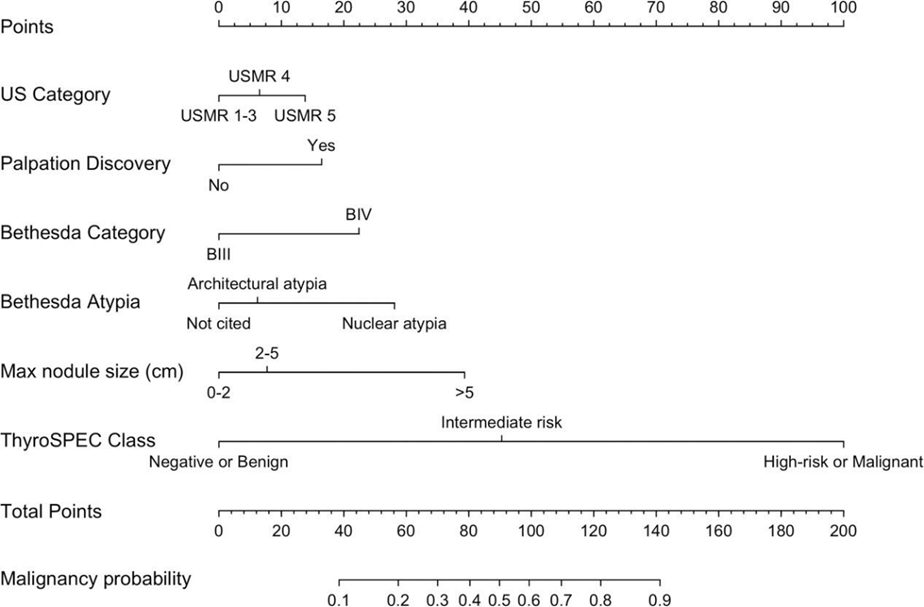
Range of ThyroSPEC™ Test Performance as calculated by including only resected nodules and by including both resected and unresected nodules that are assumed benign with at least one year follow-up and stable/improved ultrasound (3 year ThyroSPEC™ outcomes Wu et al., Thyroid, 2025):
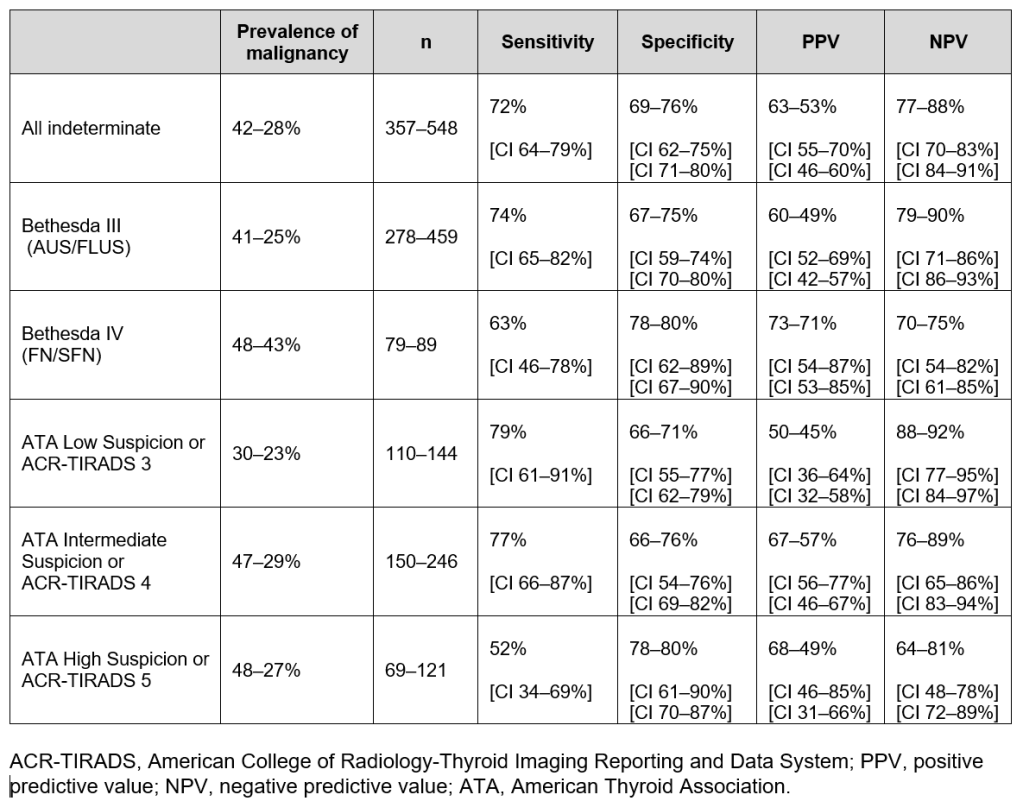
Revised Thyroid Nodule Molecular Diagnostic Test Results: Interpretation and Management Guide (implemented April 2025). Range of ThyroSPEC™ ROM as calculated by including only malignant cases defined as nodules that were malignant on surgical histology and by including both resected and unresected nodules that are assumed benign with at least one year follow-up and stable/improved ultrasound (3 year ThyroSPEC™ outcomes Wu et al., Thyroid, 2025):
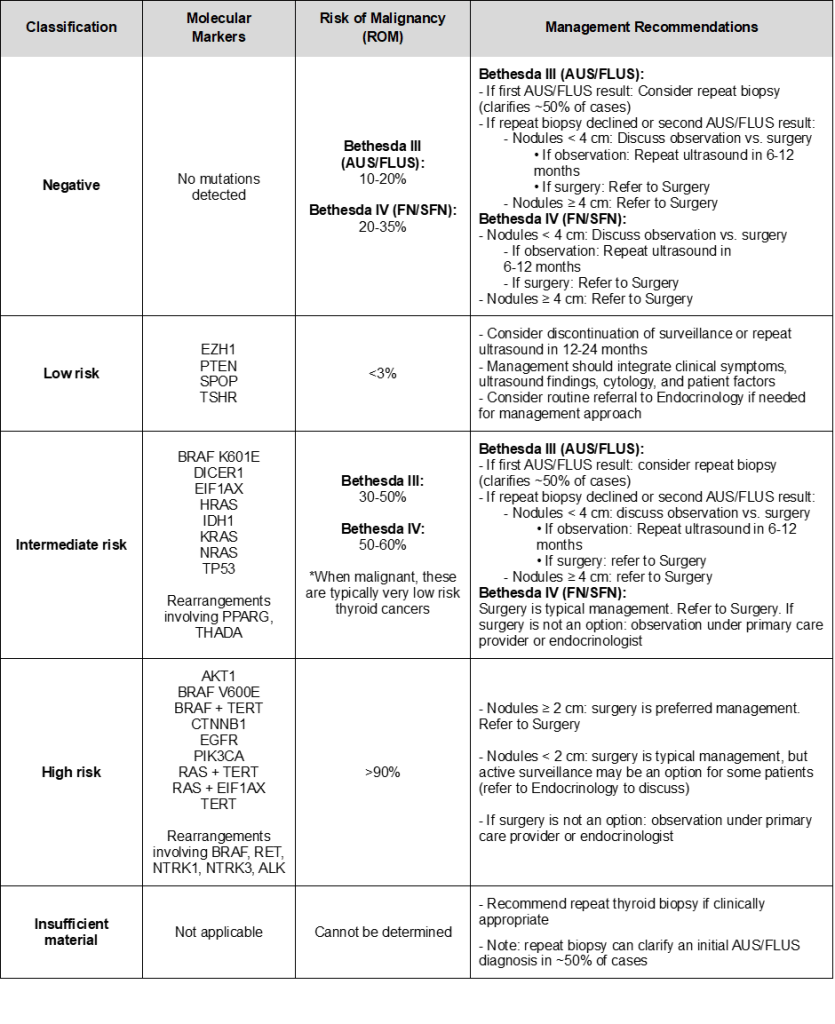
For no mutation detected and intermediate risk mutations, please see the nomogram for an integrated malignancy risk assessment based on Calgary data (Wu et al., Thyroid, 2025):
The integrated malignancy risk stratification of intermediate risk mutations allows to select 40% of RAS-positive nodules for successful active surveillance (Wu et al., Thyroid, 2026).
Using ThyroSPEC™ for Bethesda III and IV nodules in the context of an interdisciplinary lobectomy proposal and in the setting of an optimized thyroid nodule diagnostic pathway, the number of completion thyroidectomies can be reduced by increasing the number of appropriate upfront total thyroidectomies (Yeo et al., European Thyroid Journal, 2026).
An optimized thyroid nodule diagnostic pathway — including reflexive ThyroSPEC™ testing of Bethesda III (AUS/FLUS) and Bethesda IV (FN/SFN) nodules and less aggressive surgical management performed by more high-volume thyroid surgeons — is associated with improved ultrasound malignancy risk assessment reporting quality, a higher rate of reported Bethesda atypia, and a greater prevalence of malignancy among resected nodules (Wu et al., 2025 Annual Meeting of the American Thyroid Association, Scottsdale, AZ).
Further clinical impacts of ThyroSPEC™ testing are:
- significant decrease in repeat FNAs,
- increase in risk of malignancy for Bethesda III (AUS/FLUS) and IV (FN/SFN) nodules,
- increase in diagnostic yield due to a higher malignancy rate of resected nodules,
- earlier surgery for patients with high-risk mutations: patients with high-risk mutations underwent surgery 78 days after molecular testing, compared with 184 days for ThyroSPEC™-negative patients
- high NPV for the diagnostically most challenging ATA intermediate / ACR-TIRADS 4 nodules (Stewardson et al., Thyroid, 2023).
For all categories, referral to Endocrinology or Surgery can be considered if it will aid in determining the best management approach.
For a list of providers accepting thyroid nodule referrals, visit: https://www.albertahealthservices.ca/assets/info/hp/cancer/if-hp-cancer-guide-southern-alberta-providers-thyroid.pdf
Disclaimer: Interpret the above results within the context of other clinical data such as ultrasound, with clinical management decision making according to the independent medical judgement of the responsible physician and patient preferences.
ThyroSPECTM was not created to identify germline variants, nonetheless it is possible that ThyroSPECTM will discover a germline mutation incidentally. If a germline variant is reported, referral to medical genetics may be advisable.
